 For highly ionized atoms, the lines are found in the extreme UV or x-ray region.Īs the relative intensity of the lines in an atomic spectrum varies with temperature, analysis of the lines in the spectrum of a star (say) can give an estimate of the temperature of the star’s surface (photosphere).  
The light electronic transitions in atoms produces may not be in the visual part of the electromagnetic spectrum, but for atoms that are neutral or have lost only one or two electrons (yes, ‘atomic spectra’ refers to the line spectrum of ions too!), most lines are in the UV, visual, or near infrared. Of course, for an extragalactic object – a quasar, perhaps – you need more than one line to make a certain identification … because the universe is expanding (and so you don’t know how much just one line may have been redshifted). From the Lyman series to the Pfund series, the wavelength increases from 1000A o to 50,000A o.As the atomic electron energy levels are unique to each element, the lines in a spectrum (emission or absorption) can be used to identify the elements present in the source (a star, say) or gas between the source and us (e.g. In general, the wavelength of emission may be estimated using the Balmer equation. Photons emitted from the atoms in their excited states strike the prism and separates the light. the difference between higher and lower energy levels, five types of spectral series are found for hydrogen.īecause the hydrogen atom has an infinite number of unoccupied orbits, the excited electron can return to 1,2,3,4, or 5 orbits, giving birth to the Lyman series, Balmer series, Paschen series, Bracket series, and Pfund series. The instrument used to view line spectra is a spectrometer. Based on the wavelength of energy emitted, i.e. Because the excited electron is not stable, it returns to lower energy levels by emitting radiations in jumps, giving rise to line spectra. When hydrogen is subjected to an electric discharge at high voltage and low pressure, the molecules are first dissociated into atoms, and then electrons are promoted to higher energy levels depending on the amount of energy absorbed. Hydrogen gives absorption as well as emission spectra. Electrons in these orbits may absorb energy, be promoted to higher energy levels, and then radiate the absorbed energy at a characteristic wavelength. An atom, according to Bohr’s atomic theory, has multiple orbits with different energies. The hydrogen atom produces five series of line spectra that can be described using Bohr’s atomic theory. When absorption and emission spectra of the same element are compared, the dark lines of absorption and bright lines of emission spectra appear at the wavelength.Įxplanation of hydrogen spectra on the basis of Bohr’s atomic model A spectroscope is used to record these spectra. As a result, these spectra are effective tools for identifying elements. The emitted radiations resulted in a series of bright lines on a dark background, which is known as an emission spectrum.Įach line in the absorption or emission spectra corresponds to a specific wavelength of radiation, therefore no two elements have the same pattern of spectral lines in the absorption or emission spectra. When the heating is turned off, the atoms revert to their ground state by emitting radiation of a specific wavelength. 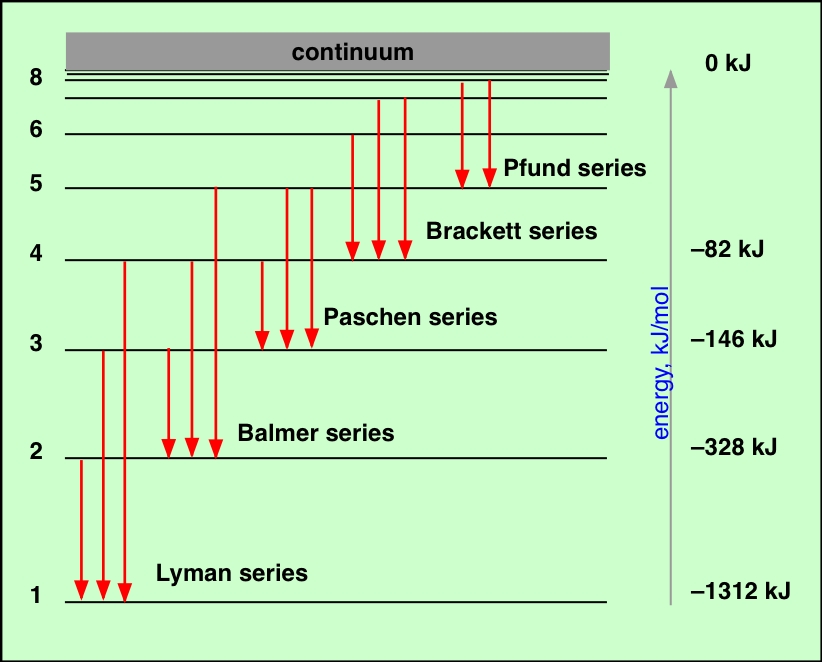
When an atom in its vapor state is heated to a high temperature, the electrons get excited. This book discusses regularities in spectra emitted by monatomic gases. This behavior of atoms produces a series of dark lines on a background called atomic absorption spectra. Atomic Spectra compiles papers on the highlighted developments in the atomic spectra. The light of other wavelengths gets transmitted. When an atom in its vapor state is placed in white light, it absorbs light of characteristic wavelength due to the electrons present in it. There are two types of atomic spectra: Absorption spectra A dark area separates these spectral lines from one another. Due to their distinct orbitals or energy levels, all atoms produce line spectra or discontinuous spectra. The spectra, in fact, are energy deals of electrons. An atom’s electron can absorb or radiate the energy of a specific wavelength, which can be recorded as atomic spectra in the form of bands or lines. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
